This month, we highlight the recent work of several students from the Balasubramanian Lab at the University of Cambridge using the EvoluChem™ Photoredox Boxes in their graduate studies. Shankar Balasubramanian is a godfather in the field of nucleic acid chemistry and methods to sequence epigenetic modifications. In recent years this work has involved visible-light applications. Ben Mortishire-Smith, a Ph.D. student in the Balasubramanian lab, wrote us to explain that he and his colleague Matt Simpson have “racked up many hundreds of hours running the PhotoRedOx Boxes since we got them in 2019.” Matt included that the 365nm lamp “saved his Ph.D.” With dedication like that, we would certainly like to share their story.
Two recent papers from the Balasubramanian lab using EvoluChem™ PhotoRedox Boxes and LEDs caught our attention. First up is A Photoredox Reaction for the Selective Modification of 5-Carboxycytosine in DNA published last month in JACS (Open Access) by authors Benjamin J. Mortishire-Smith, Sidney M. Becker, Angela Simeone, Larry Melidis, and Shankar Balasubramanian. Once again in the words of Ben, he states “we use a blue-light iridium-catalysed reaction, which enables 5-carboxycytosine and 5-methylcytosine modifications in DNA to be sequenced”. If you aren’t entirely certain how a blue LED is going to help you sequence DNA modifications, don’t be ashamed we were confused as well. So, let’s try to figure this out together.
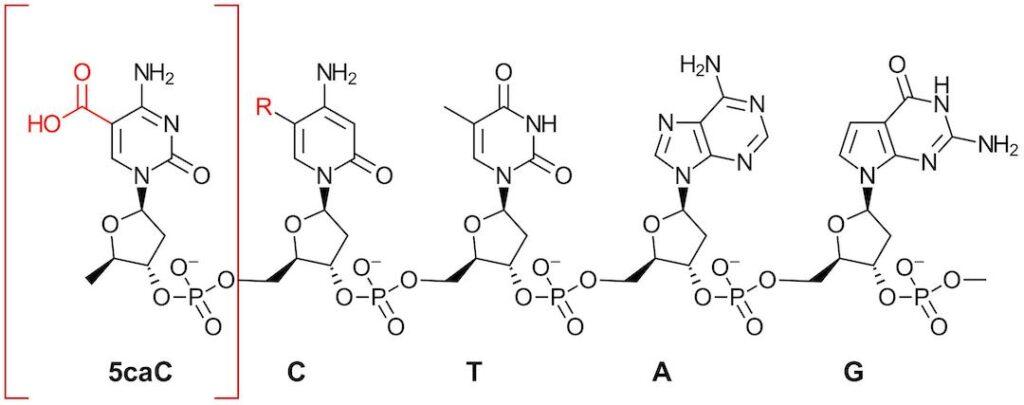
We know that there are 4 nucleobases, adenine (A), cytosine (C), guanine (G) and thymine (T) and that modification of these 4 bases represents the epigenetic code. As the authors state, methylcytosine (5mC) is the most abundant mammalian modification (5% of cytosine sites). Further oxidation of (5mC) results in 5-hydroxymethylcytosine (5hmC), 5-formyl-cytosine (5fC) and 5-carboxycytosine (5caC). We will gloss over the myriad of reasons understanding these modifications are important for either living or dying, but if you want to understand how something works, it’s important to understand what it is to start. And for that you need to be able to differentiate between all these modifications. In this work, the authors focus on 5caC which is poorly understood with relatively low abundance (fewer than 1 base in 106)
Figure 1: Selectively Targeting carboxylic acid in 5caC in DNA. R=H, Me, CH2OH, CHO (Figure adapted from Fig 1 in Ref1)
Visible-light photochemistry is mild, controllable, tunable, and selective (all the buzzwords you want to hear when finding a useful reaction condition). So, it makes sense that visible-light photocatalysis is a method of choice when trying to differentiate between very similar type molecules like the DNA nucleobases shown in Figure 1. Here the authors ingeniously target the carboxylic acid group in 5caC. Photocatalyzed decarboxylation is well established method for eliminating CO2 and generating a carbon-centered radical. And while shining a bright blue light on some DNA seems like the origin story in a bad superhero movie, it also may have some scientific benefits as well.
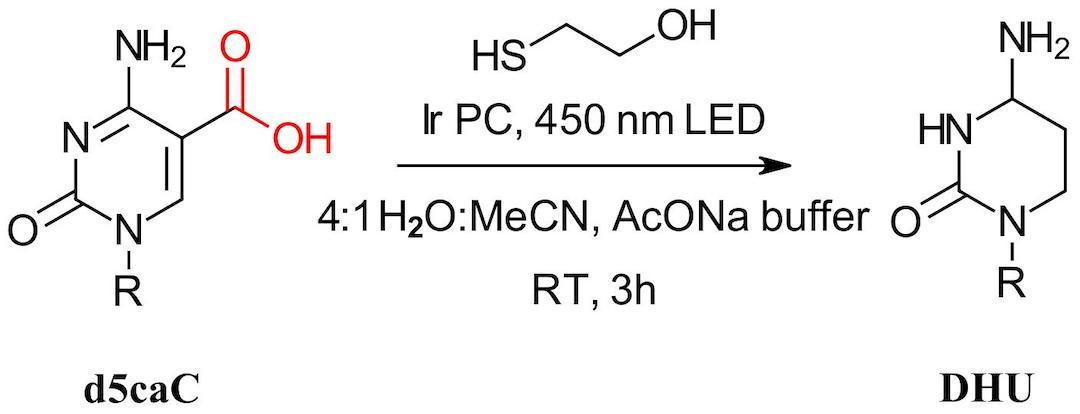
Figure 2: Photochemical conversion of 5caC to DHU
Using an iridium catalyst, 2-mercaptoethanol, acetate buffered water:acetonitrile and 450 nm LEDs in a Photoredox box, the authors observed near quantitative conversion of a single nucleotide 5caC mimic to DHU in 3 hours (Figure 2). The product demonstrated a loss of 41 Da by HRMS, loss of absorbance at 260 nm and with the DHU structure ultimately being confirmed by NMR. The selectivity of the reaction was then confirmed by treating an array of nucleosides and modified bases to the photochemical conditions with minimal change observed. Moving on to DNA oligonucleotides, the authors prepared a 10-mer containing 1 5caC base and observed the -41 mass change by MS within 10 minutes. No off-target modifications were observed.
Not content, they moved onto a 74 base sequence containing 1 5caC and bacteriophage genomic DNA with 5caC modifications and implemented this reaction into a next-generation sequencing workflow. Following photochemical treatment, 5caC is converted to DHU which can be read by DNA polymerases to incorporate an adenosine base and thus a T by PCR. Ultimately converted 5caC positions are read as C-to-T mutations. For the 74 base sequence 82.6% efficiency was observed for 5caC modification with 0.29% unmodified cytosines. In the bacteriophage system 85% efficiency was observed. Ultimately, the authors have demonstrated the first direct modification of a DNA base via photochemistry and a useful system for finding 5acC epigenic modification. We can’t do justice to the full explanation of the next-generation sequencing workflow and suggest that you check out the full work.
The second paper is Selective Functionalisation of 5-Methylcytosine by Organic Photoredox Catalysis Published in Angewandte Int. Ed. (Open Access) by authors Mathew M. Simpson, Ching Ching Lam, Jonathan M. Goodman and Shankar Balasubramanian. Not content with modifying 5caC, the Balasubramanian lab clearly wants to modify all our bases. Next up 5mC. All the necessary caveats from above apply, methods to identify epigenetic changes are important and it’s no less impressive that visible-light photocatalysis can selectively modify a specific DNA base in the presence of unimaginable complexity. In fact, it’s more impressive demonstrating with the right photocatalyst, reaction condition and wavelength seemingly any base may be selectively modified.
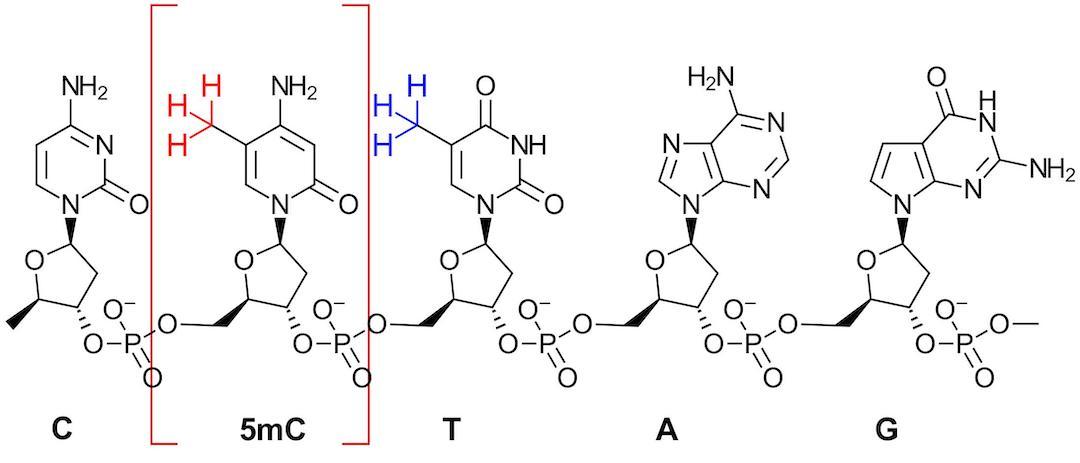
Figure 3: Selectively Targeting methyl group in 5mC for photochemical labeling in the presence of T in DNA. (Figure adapted from Fig 1 in Ref 2)
For 5mC, the challenge is successfully labeling the methyl group (red) in the presence of the incredibly similar methyl (blue) found in thymine (T) (Figure 3). As the authors state, the bond dissociation energies differ by less than 5 kJ mol-1 making direct hydrogen-atom abstraction unfavorable. However, the T nucleobase has 0.53 V higher redox potential than 5mC suggesting that they just need to find the right photosensitizer to promote single electron transfer and the right trapping agent.
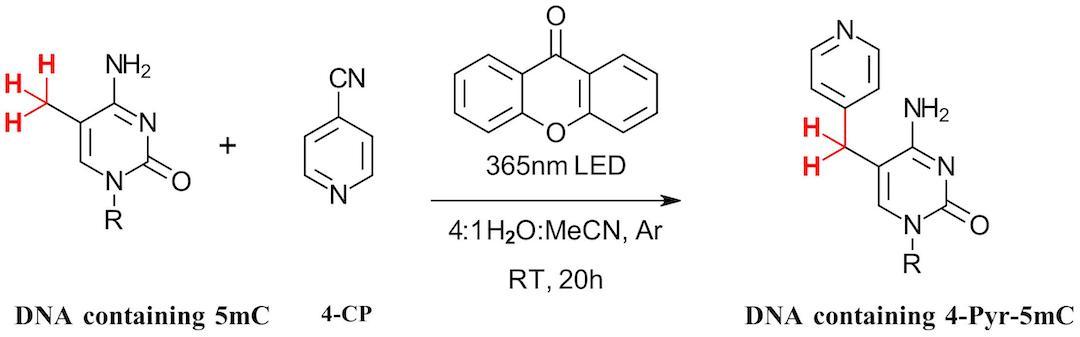
Xanthone was selected along with the suitable 365 nm LED. Acetonitrile helps to solubilize the catalyst. The reaction was performed under Ar to exclude oxygen and prevent oxidation of 5mC. In this condition, 42% of 4-Pyr-5mdC was observed after 20 hours. The reaction was then screened on the other common DNA nucleosides. Only 3% labeled dT was observed while the remaining bases were inert to the photocatalysis condition. Following this, a series of 4-CP derivatives were screened for labeling 5mC. 3-methyl-4-cyanopyridine increased the conversion to 58% while several other derivatives were suitable alternatives.
Figure 4: Photochemical oxidation of 5mC
The photochemical labeling reaction was transferred to 12-base oligonucleotides containing 5mc with notable modification of the reaction condition, including doubling catalyst concentration, buffering reaction at pH 7.1 and lowering reaction temperature at 4°C. Here 91% of the nucleotide was observed at 2 hour with pyridination accounting for 48% of the products. Alternate products include Minisci style coupling next to the pyridine nitrogen in 4-CP. Following this result, the authors also further investigated the reaction mechanism, tested additional DNA sequences and transferred the reaction for use in RNA. Altogether, another nice example using photocatalysis to selectively modify DNA bases. No doubt there will soon be a Balasubramanian toolbox of photocatalysis condition for modification of all the epigenetic markers. We look forward to seeing them all.
If you have a story that you would like to share about using HepatoChem™ photochemistry equipment, send us a note and we would be happy to highlight it in a future newsletter. For our previous articles, check out our blog.
REFERENCES:
(1) Mortishire-Smith, B. J.; Becker, S. M.; Simeone, A.; Melidis, L.; Balasubramanian, S. A Photoredox Reaction for the Selective Modification of 5-Carboxycytosine in DNA. J. Am. Chem. Soc. 2023, 145 (19), 10505–10511. https://doi.org/10.1021/jacs.2c12558
(2) Mathew M. Simpson, Ching Ching Lam, Jonathan M. Goodman, and S. B. Selective Functionalisation of 5″Methylcytosine by Organic Photoredox Catalysis. Angew. Chem. Int. Ed. Engl. 2023. ASAP. https://doi.org/10.1002/anie.202304756