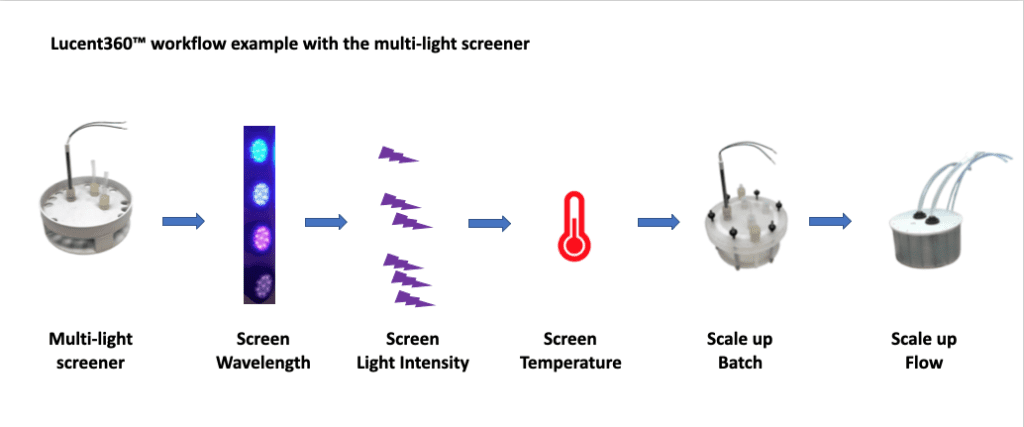
A few weeks ago, we discussed the history of the Lucent360™, our new photoreactor for light and temperatures control for screen to scale, in both batch and flow. This month, we want to share a series of experiments that demonstrate the value of the Lucent360™ for studying a photochemical reaction. This project was brought to us by chemists at the IRCOF and Oril in France*. There is no shortage of possibilities for how to best study a photochemical reaction. Having the tools available to design the best experiment is key. We understand that everyone’s workflow will be different and there is likely no correct order for running a set of reactions. However, here a few steps for taking a new reaction from the screening scale to both batch and flow in the Lucent360™.
Figure 1: Example Workflow Using Lucent360™
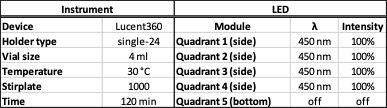
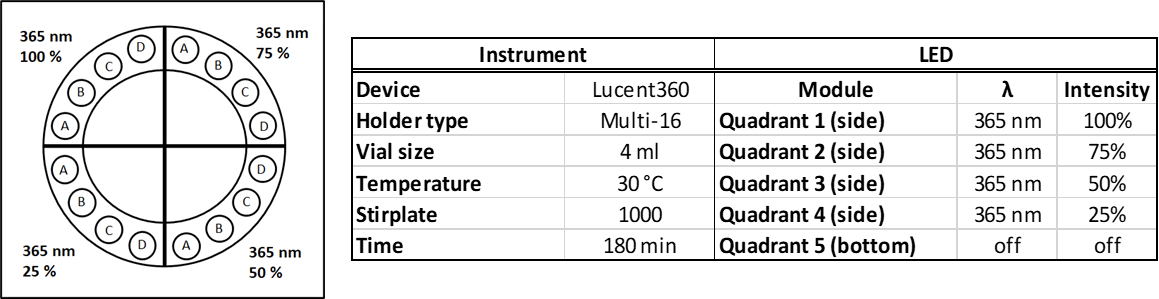
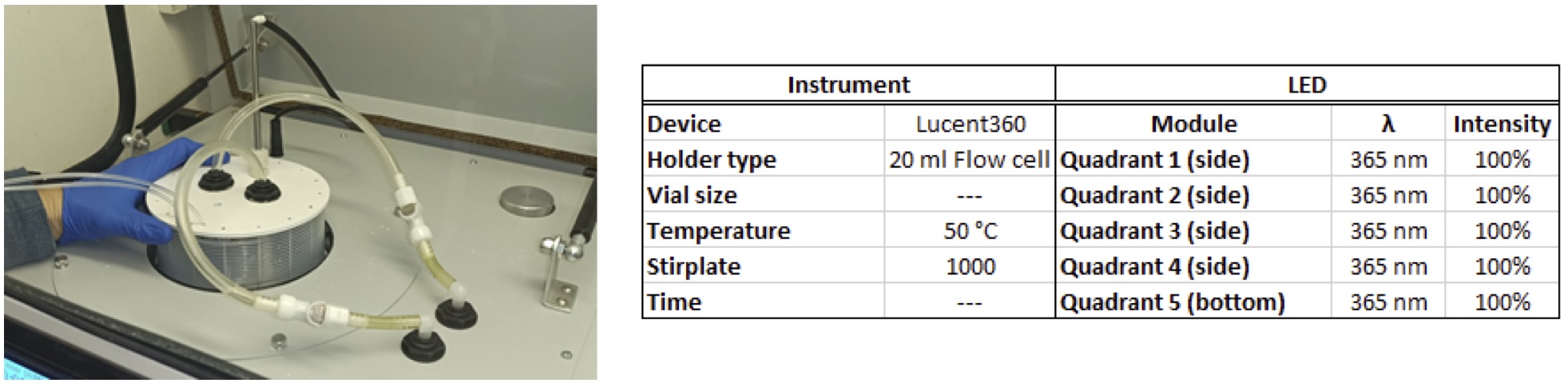
 The Lucent360™, is designed with 5 interchangeable LED modules (4 side modules and 1 bottom module) to fully immerse a temperature-controlled chamber with light. Each module can operate at either 365 nm, 380 nm, 405 nm and 450 nm (with many more options to come) with variable light intensity. Interchangeable vial holders for reactions vials from ranging from 0.3 ml up to 20 ml fit in the reaction chamber and allow you to screen 16 to 48 reactions at a time (depending on the vial size). Select the temperature you want and keep your reaction constant from 0 °C to 80 °C. A typical experiment with a standard holder might be something that looks like this: (4 side 450 nm LEDs set at 100% intensity, with the reaction chamber set at 30 °C, 24 vials total, each @ 4 ml each containing a set of test substrates with 24 different catalysts (Table 1).
The Lucent360™, is designed with 5 interchangeable LED modules (4 side modules and 1 bottom module) to fully immerse a temperature-controlled chamber with light. Each module can operate at either 365 nm, 380 nm, 405 nm and 450 nm (with many more options to come) with variable light intensity. Interchangeable vial holders for reactions vials from ranging from 0.3 ml up to 20 ml fit in the reaction chamber and allow you to screen 16 to 48 reactions at a time (depending on the vial size). Select the temperature you want and keep your reaction constant from 0 °C to 80 °C. A typical experiment with a standard holder might be something that looks like this: (4 side 450 nm LEDs set at 100% intensity, with the reaction chamber set at 30 °C, 24 vials total, each @ 4 ml each containing a set of test substrates with 24 different catalysts (Table 1).
Table 1: Lucent360™ experiment parameters for a typical experiment:
Here you can quickly answer the question, which catalyst works the best? Or maybe you mix and match a screen or solvents, or bases, or an additive. You may already own a device that can handle a screen like this (if you are controlling the temperature, then maybe you have our PhotoRedoxBox TC). If this was the extend of what we wanted to do, then we would have never gone to the effort to build the Lucent360™. But there are so many other experiments that you might want to try that end up being a significant amount of work. So maybe you don’t ever get around to trying them.
Multi-light screener experiments
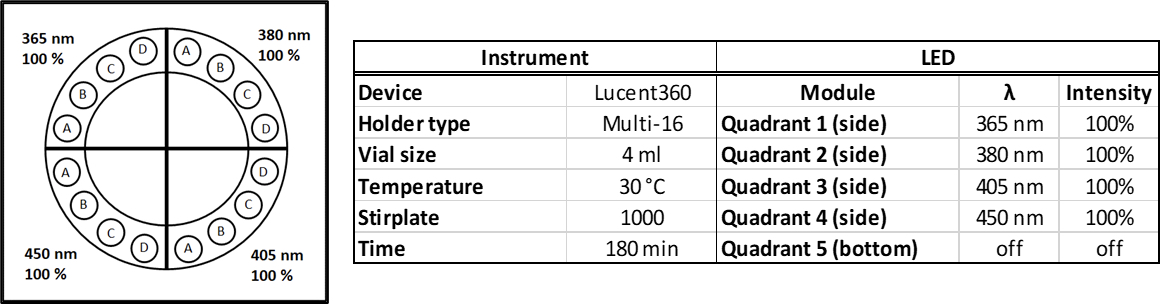
It is the multi-light screener holder (Figure 2) that unlocks the power of the Lucent360™. The multi-light screener separates the light from each quadrant allowing each module to operate independently with a different wavelength or light intensity under the same temperature and experimental conditions. Instead of running a full set of experiments over and over again, to change different parameters, get more information out of each screen.
Figure 2: Multi-light screener
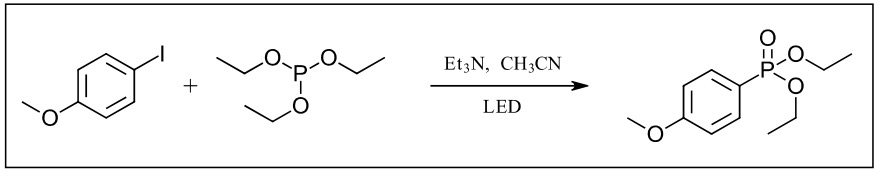
 We set out to investigate this photochemical version of the Arbuzov reaction, the reaction of an alkyl halide and a trialkyl phosphite to make an alkyl phosponate. In our example, the reaction of iodoanisole and triethylphosphite and triethyl amine (Figure 3).This project was brought to us by chemists at the IRCOF and Oril in France as a reaction that was originally performed in flow.
We set out to investigate this photochemical version of the Arbuzov reaction, the reaction of an alkyl halide and a trialkyl phosphite to make an alkyl phosponate. In our example, the reaction of iodoanisole and triethylphosphite and triethyl amine (Figure 3).This project was brought to us by chemists at the IRCOF and Oril in France as a reaction that was originally performed in flow.
Figure 3: Photo-Arbuzov reaction in Lucent360™
Step 1: Screen Wavelength
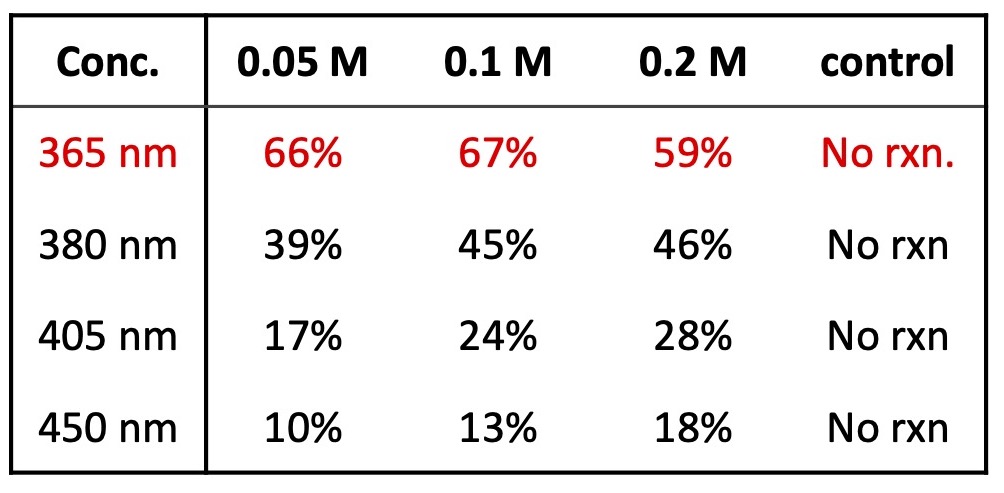
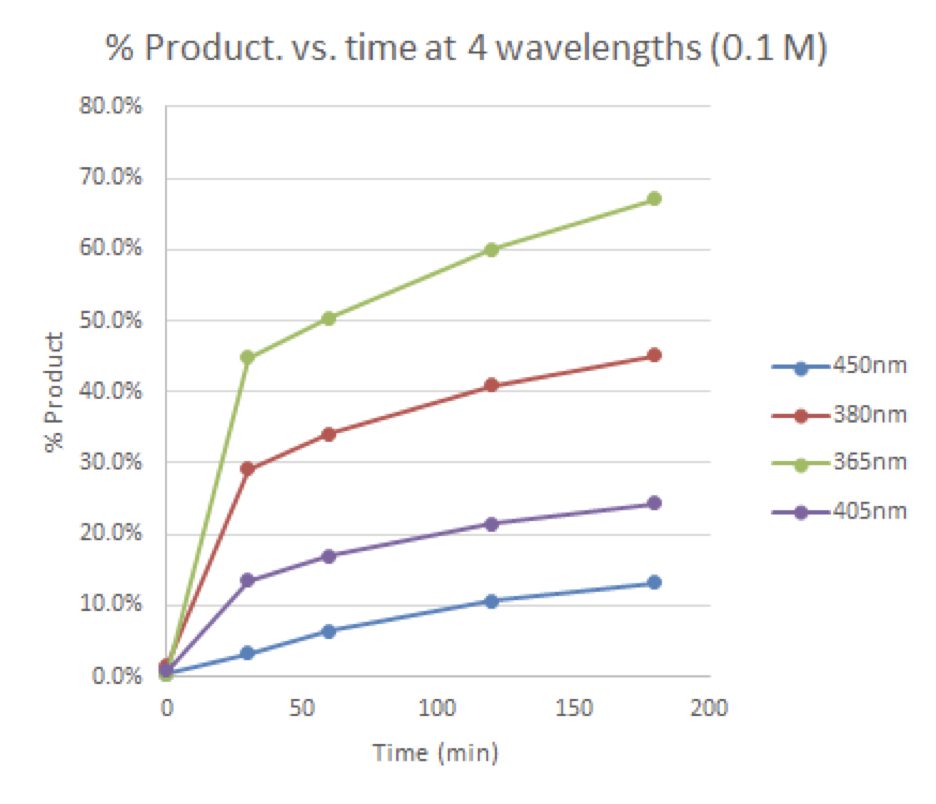
To start, what wavelength and concentration should we run this reaction? With the Lucent360™, it is straightforward to test multiple wavelengths in a single experiment. For example, screen 16 conditions – 4 wavelengths, 4 concentrations in one experiment with temperature control.
Here, the reactions were performed in Lucent360™ equipped with 4 different side light modules (365 nm, 380 nm, 405 nm, 450 nm) set at 100% intensity using the multi-light screening module. Each reaction was prepared in a glove box with sparged acetonitrile in 4 ml crimp cap vials with 2×7 mm stir bars. In each quadrant, were four experimental conditions investigating 3 concentrations of the 2 coupling components (0.05M, 0.1M, 0.2M iodoanisole) and a control without triethylphosphite. (p-iodoanisole (1 equiv.), triethylphosphite (2 equiv.), triethylamine (2 equiv.), in 2 ml ACN). For all experiments, the products were identified by LC-MS with quantification by DAD in comparison to calibration standards of anisole diethylphosphonate.
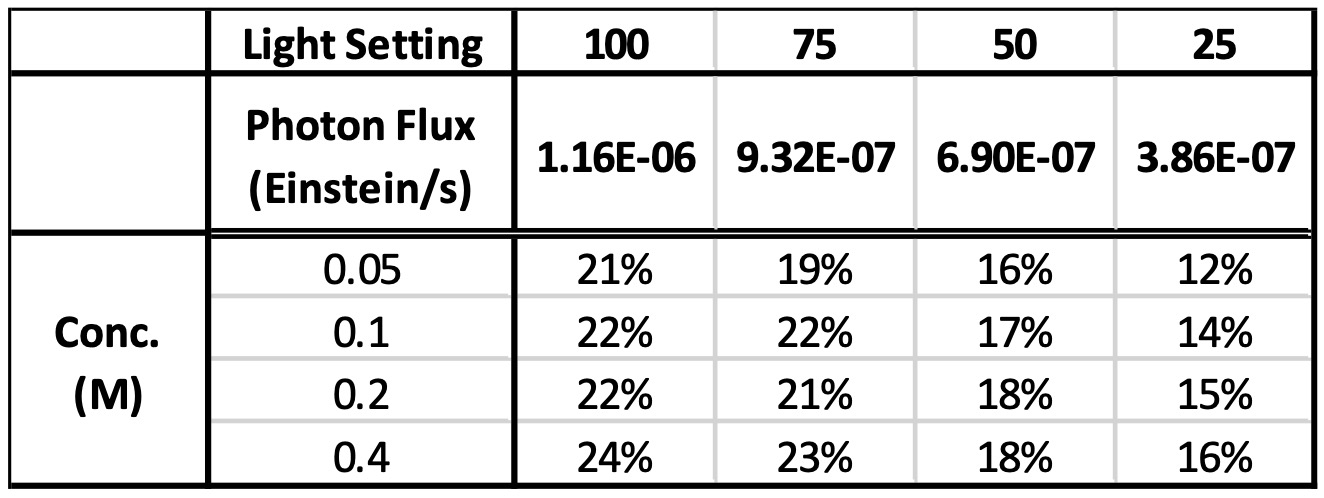
Table 2: Wavelength screen (% product at 180 min) with 100% light intensity
Figure 4: Time course for 0.1 M reactions vs. wavelength
Step 2: Screen Light intensity
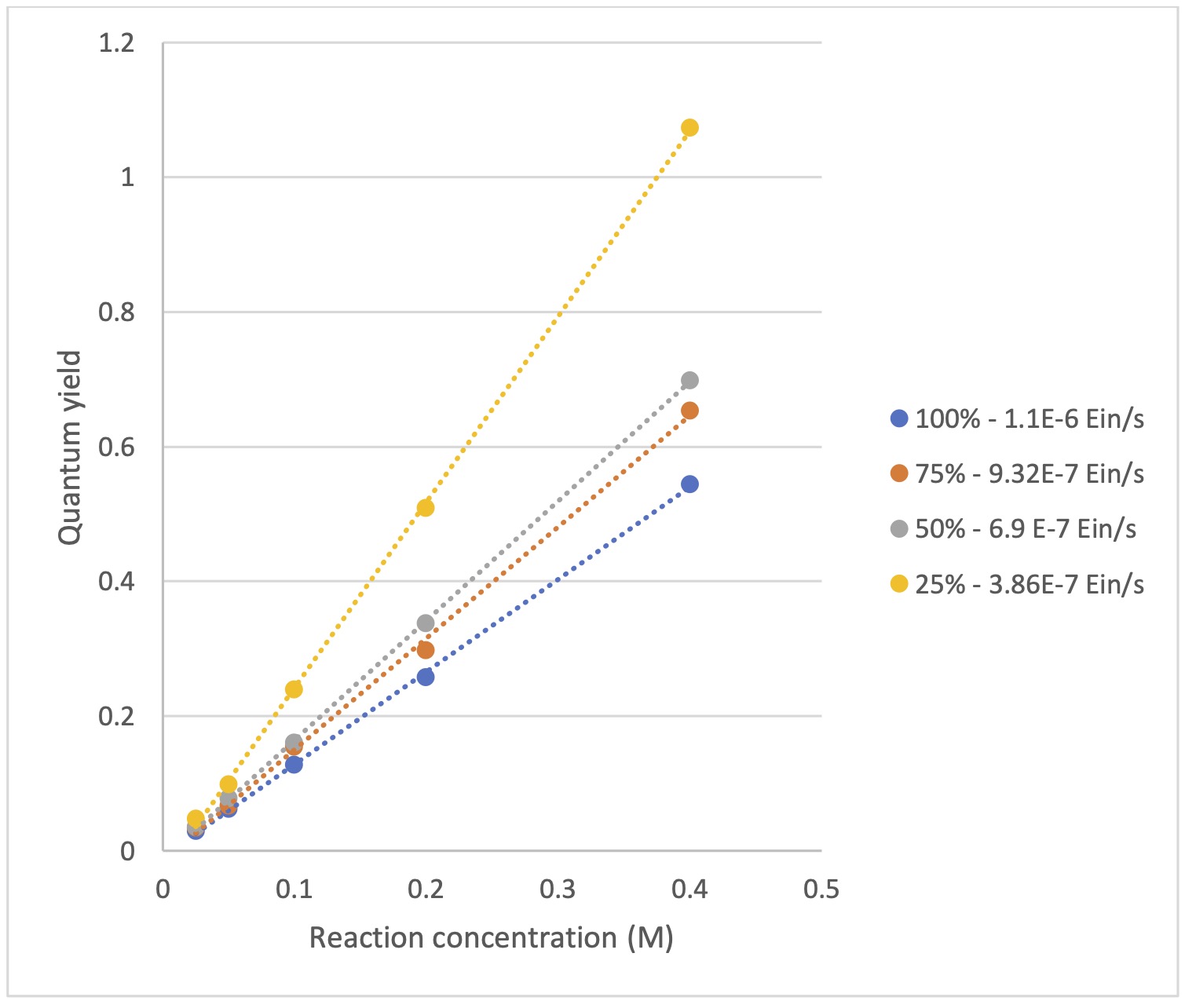
After selecting 365 nm as our wavelength, how much light do we need for our optimal reaction? Too little light, and we may not be optimizing our reaction rate. Too much light might be detrimental for a host of reasons, such as instable products, or starting materials, unwanted side reactions, or simply to limit the amount of cooling required to keep the reaction temperature constant. Next, we screen 16 reactions (four reaction concentrations 0.05 M, 0.1 M, 0.2 M, 0.4 M) in the Lucent360™ equipped with four side 365 nm modules set at 25, 50, 75, 100% intensity using the multi-light screening module. The photon flux for each intensity was determined by chemical actinometry according to the method described previously. For this case, in the Lucent360™ each quadrant corresponds to the following photon flux (3.9×10-7 Einstein/s, 6.9×10-7 Einstein, 9.3×10-7 Einstein/s and 1.1 x10-6 Einstein/s,).
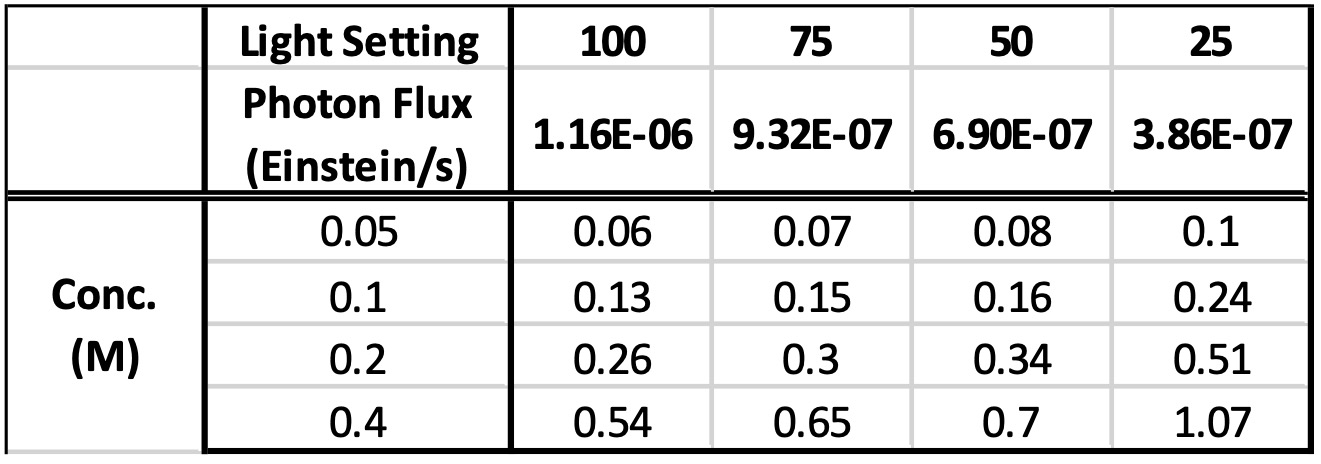
There are many levels to the data that can be acquired in one set of experiment like this, (final product, impurities, remaining starting material, initial rate of the reaction, and quantum yield). And we’ve plotted it all. What stood out for us in this data is an interesting effect when we use the initial rate of each reaction, along with the chemical actinometry data to determine the quantum yield at each concentration and light intensity. For each light intensity, the quantum yield has a linear relationship with the reaction concentration (see Figure 5). What do we think this tell us? We’re not entirely sure, but we’ll writing more about this for additional reactions shortly.
Table 3: Product formation at 5 min with varying light intensity
Table 4: Quantum Yield of each reaction determine at 5 minutes
Figure 5: Quantum yield vs. Reaction concentration based on initial rate (5 min)
Step 3: Screen Temperature
Next, we used the temperature control portion of the instrument to monitor this reaction at temperatures from 10 °C up to 50 °C with a few additional controls. We used the Lucent360™ equipped with four 365 nm side light modules at 100% intensity and monitored the time course of each reaction. Here, we observed very little difference in product formation vs. temperature for both the initial point and at 300 minutes, however, higher temperature (40 and 50 °C) had significantly higher conversion at 120 and 240 minutes.
Table 5: Effect of Temperature on reaction
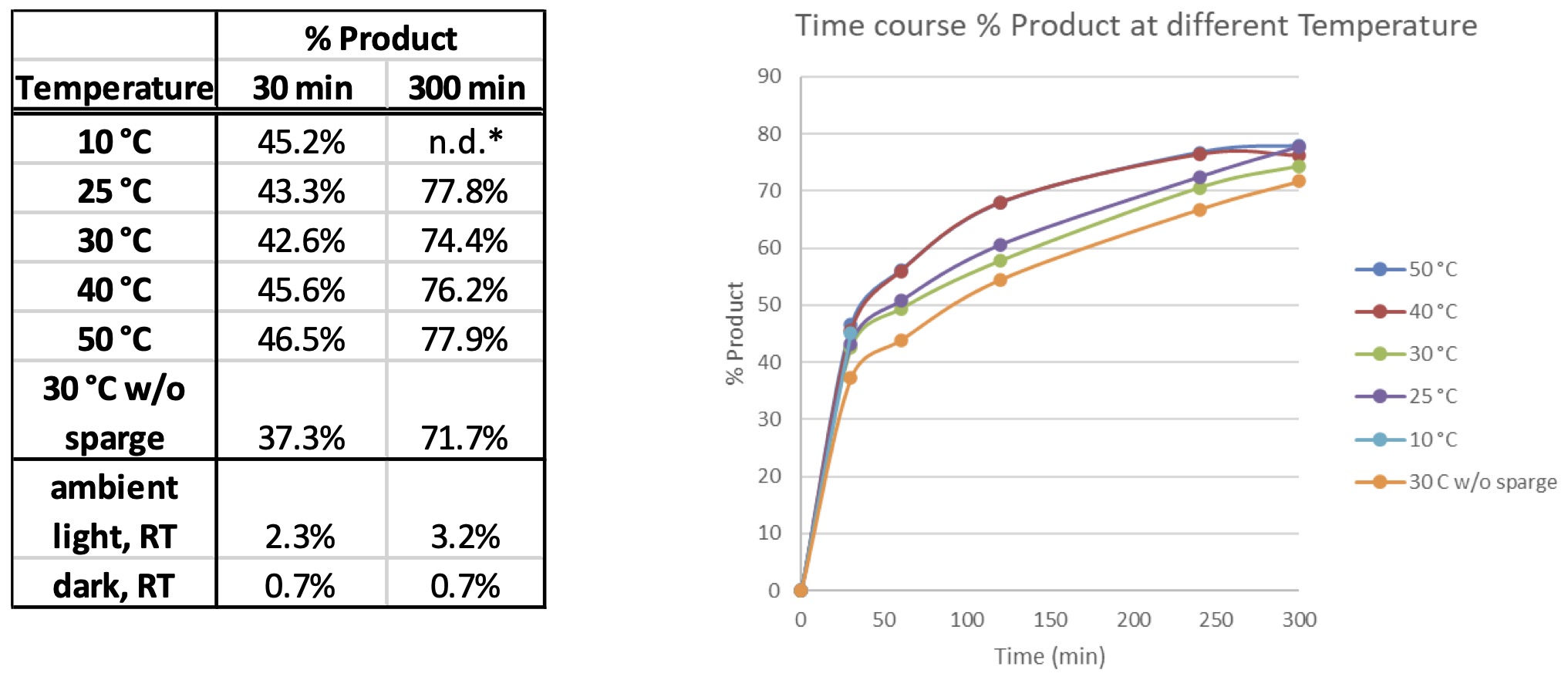 *Only initial time point determined for 10 °C experiment
*Only initial time point determined for 10 °C experiment
Step 4: Scale up in Batch
Screening is nice, but for most people the end goal is a reaction that can be scaled to produce a useful amount of material. With the Lucent360™, the same instrument that you used to screen your reaction can be directly used to scale up the reaction in batch. We tested our phosphonylation reaction in our Lucent360™ 700 ml reactor vessel. The reaction was prepared in the 700 ml reactor (includes inlet and outlet port to cool reaction, gas line in/out and a temperature probe to monitor the reaction) and transferred to Lucent360™ and sparged with N2.
The reaction was performed with 5 light modules (365 nm) at 100%, 50 °C at 500 ml scale and 23.4 g p-iodoanisole corresponding to the 0.2 M concentration condition. Did we need to go up to 50 °C based on previous data? We did not. Did we want to demonstrate an example of scaling up a reaction to 500 ml, with the lights full blast and the heat control on and maintain the temperature for the full run of the experiment? We did. And we did. We monitored the reaction and observed:
- 56% product at 5 hours (32.9% starting material remaining) – ~13.7 g product formed in 5 hours.
- 68% product at 22 hours (17% starting material remaining) – ~16.6 g product formed in 22 hours.
Step 5: Scale up in Flow
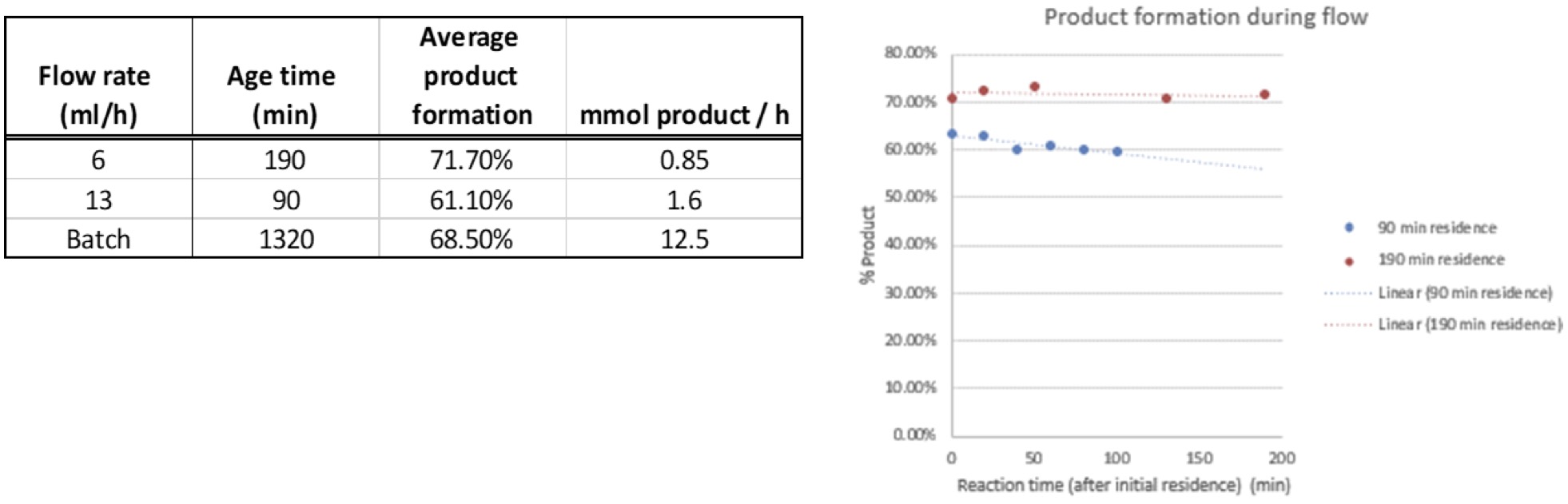
If the reaction works in batch? How does it look in flow? We took the parameters that we established from the batch reactions, along with some previously reported residence times from IRCOF/Oril for this reaction in flow and tested our Lucent360™ 20-ml flow cell.
To start, we prepared a 40 ml mixture of the 0.2 M scale reaction in sparged acetonitrile. The reaction was performed at 50 °C in the Lucent360™ with 365 nm light (5 quadrants) at 100% light intensity with a 20 ml flow cell. The flow rate set was set at either 6 ml/hour (190 min residence time) or 13 ml/hour (90 minute residence time). Aliquots taken through the course of the run and diluted into DMSO for analysis. Similar conversions were observed for the flow reactions in comparison to the batch reaction. However, for this specific reaction, the long residence time makes the throughput in batch in significantly higher for batch than flow.
Figure 6: Comparing batch and Flow
Summary
With the Lucent360™, using the interchangeable LED modules, vial holders and multi-light screening tool, it is possible to identify important information regarding the reaction parameters needed to scale a reaction to both batch and flow. Can smarter people than us design experiments to take advantage of the Lucent360™? We hope so. Can you use data like this for your favorite reaction? We hope that as well. In the coming weeks, we’ll be adding more case studies using the Lucent to our website at hepatochem.com.
* Bruno Schiavi and Damien Thevenet, Oril Industrie – Bolbec – Normandy – Affiliated with « Les Laboratoires Servier », France
Philippe Jubault, Romain Lapierre, Thi Minh Thi Le and Thomas Poisson, IRCOF, COBRA Laboratory UMR6014, Rouen, Normandy, France