Photochemistry Surprises!!!

The Lucent360™ is the most complete photochemical reactor available for controlling and screening reaction parameters like wavelength, light intensity, reaction time, and temperature at both small and large scale. But specs only matter when you can see how the instrument fits into a real research and development program. A recent paper from GSK shows exactly that.
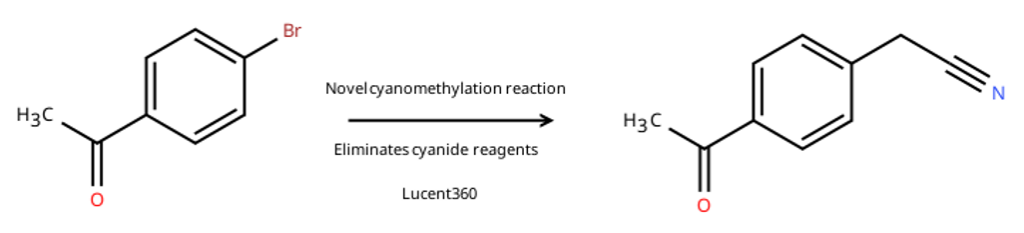
In Chemistry: A European Journal, lead author Gemma Cook and coworkers report how the Lucent360™ helped them discover a new reaction and remove a highly toxic cyanide reagent from their synthesis. The paper is open access: “Expedient Discovery of a Metallaphotoredox Cyanomethylation for Synthesizing α-Aryl Nitriles.”
Why α-aryl nitriles matter and why they’re hard to make
Several marketed drugs, including Verapamil, Ariflo, and Anastrozole, contain the α-aryl nitrile motif. The nitrile group is small enough to fit tight binding pockets while still forming productive interactions with a target — and once installed, it serves as a synthetic handle for accessing amines, carboxylic acids, tetrazoles, and more.
The catch: most existing methods to install this group rely on highly toxic cyanide reagents that are difficult to use at industrial scale, or require harsh conditions that limit functional group tolerance. A milder route would unlock library-scale SAR work on α-aryl nitriles.
A serendipitous discovery
While attempting a different photochemical alkylation under standard Ir/Ni conditions, the GSK team observed an unexpected product: cyanomethylation, with the acetonitrile solvent itself trapping the aryl bromide. That accident became the starting point for a brand-new method.
What followed was an extensive HTE and Design-of-Experiments campaign — optimizing photocatalyst, nickel source, ligand, base, and stoichiometries — that lowered iridium and nickel loadings without sacrificing yield.
The Lucent360™ setup
The optimized conditions were applied to a substrate scope of medicinally relevant aryl bromides using the Lucent360™ in screening mode:
- 24 × 4 mL vial holder
- 5 independent light modules at 450 nm
- Reaction temperature held at 40 °C via thermostatic bath
- 22–88 hour irradiation
The reaction tolerated a wide range of functional groups — amides, alcohols, phenols, sulfonamides, ethers, and ortho-substituted systems — at modest but synthetically useful yields.

From discovery to drug intermediate
The team then applied their new method to a Senexin derivative, a known CDK8/19 inhibitor scaffold relevant to oncology research. The published route requires three steps, methylation, benzylic bromination, and SN2 displacement with cyanide to deliver a key intermediate. Direct photochemical cyanomethylation accomplishes the same transformation in a single step, at a comparable 29% yield, and without a toxic cyanide reagent in sight.
The authors call their work “expedient,” and that word fits. Given the right tools, a serendipitous observation can become a tangible, scalable method in a single campaign.
Could the Lucent360™ accelerate your photochemistry?
If you are working on library-scale photoredox chemistry, HTE-driven optimization, or scale-up of photochemical reactions where wavelength, intensity, and temperature all need to be controlled — the Lucent360™ is the same instrument GSK used to develop this method.
Talk to us about your application — we are happy to discuss your substrates, throughput needs, and how the Lucent360™ would fit into your workflow.
More published examples using the Lucent360™:
Holm, A. R.; Wallick, R. F.; Vura-Weis, J.; Mirica, L. M. ACS Catalysis 2026, 16 (2), 1522–1532. “Illuminating the Role of Alcohol Substrate in Nickel Photoredox Catalysis via Ground State and Transient Absorption Spectroscopy.”
Lai, E. Y.; Ackermann, L.; Johansson, M. J. Chemical Science 2025, 16, 8478–8486. “A unified approach to meta-selective methylation, mono-, di- and trifluoromethylation of arenes.”
Check Our Photoreactor Suite

