A Brief History of a PhotoReactor
We write from time to time here about the topics that we find interesting or humorous in the photoredox, visible-light photocatalysis and photochemical literature (or whichever term you might like best to describe synthetic chemistry applications with light). Some of our favorites this year are this piece on potpourri catalysis and a story about microrobots. We will have more like these soon, we promise. Also, we have started putting together our year end piece for 2021 like this one we did last year.
For this post, however, we thought we would take a little break and discuss something we have been working on for quite some time, our newest photoreactor, the Lucent360™. Our most advanced photoreactor offering yet, with temperature, light intensity, and wavelength control from screen to scale in batch and flow. But first, we thought we would give a little of the history that got us to this point. Many of you are familiar with our series of photoreactors, and products that we have been offering commercially for quite a while now.
In the beginning there was light
We didn’t set out to develop photochemical reactors, or sell LEDs, or (deliver chemical kits for that matter) but now that is the majority of what we do. We were (and still are) a small little group of chemists making metabolites using metalloporphyrins and other C-H activation catalysts for our drug discovery clients. Soon we were fluorinating compounds, doing bioanalysis, and selling chemical kits.
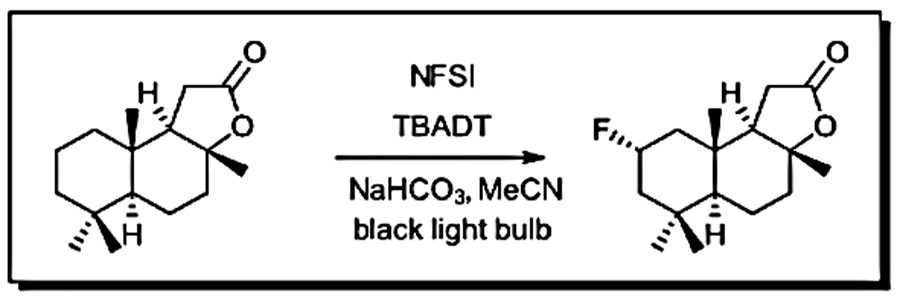
We ran our first photochemical reactions for reasons familiar to a lot of the chemists out there. We saw a new reaction in the literature that made a product that we needed to make, and it just happened to use light (see Figure 1). For us, it was this fluorination paper by Britton and coworkers (Ref 1). So, we tried it. For our light source, we had a blacklight bulb from the pet store. We taped a few vials to a stir plate and covered it with a cardboard box lined with aluminum foil. This also required making the first polyoxometalate catalyst that any of us had ever made. Some of the reactions worked well and the others didn’t at all. Often the result depended on the day of the week. When we adjusted the bulb distance or orientation, (or tried out different types of bulbs from the pet store) some reactions worked better than others. Soon after, we were running this amazing reaction from Merck with blue LEDs for the methylation of heterocycles (Figure 2).
Figure 1: Tungsten polyoxometalate fluorination as reported Britton, et. al. (2014). (Figure adapted from Ref 1)
Figure 2: Methylation of buspirone (reaction condition adapted from Ref 2)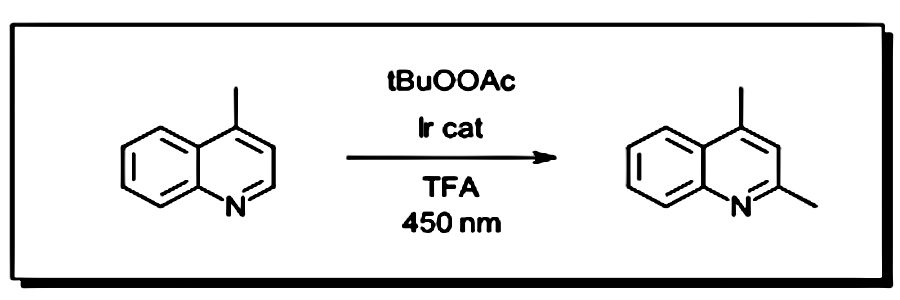
There were quite a few details that were required to set up those early photoredox reactions properly, and I think most of us were flying blind (Figure 3). As a research project, making a reaction work is part of the charm, but when you are making compounds for a service (or in MedChem) you really just want to be able to make the product you want the first time, every time. Setting up reactions with a single vial or two on a stir plate with a single lamp shining on it, simply wasn’t going to be a long-term solution.
Figure 3: An incomplete selection of light sources we tried through the years.
For our first foray into the world of building photoreactors, the solution was a plastic pipette tip box with blue LED strips and a fan. Then came a wooden box and mirrors, eventually a 3D printed design, and finally we learned about the world of plastics manufacturing (Figure 4). (Along the way we received a patent for our design (US Patent #10,906,022). So, we took our new box to do photoredox chemistry to our marketing team (yeah, it was just us). After endless discussions over drinks and bad tacos, we finally had an answer – the PhotoRedox Box – original we know. Naming things is hard. (To be fair, we initially intended to make the box out of red plastic with a logo something like this – PhotoRedox- but at the time, the only LED we had was blue 450 nm, and red plastic was more difficult to source than the blue plastic (and we liked the look of it in blue better). It turned out that many people wanted to buy our reactors and so we sold them. And then we sold a lot them and along the way changed the direction of our business.
Along the way we started manufacturing LEDs, safety screens and a variety of vial holders, based on our own ideas for the products that we wanted to use and requests from you, our customers. Some of these ideas eventually found their way for sale while some of the craziest ideas are still awaiting the time and resources to develop sufficiently. When we wanted to try a reaction at 0 °C, we designed the PhotoRedoxTC. And when we wanted to know exactly how much light was in our reaction, we learned about chemical actinometry.
All of this is to say that the products that we have designed and sold are first and foremost products that we want to use ourselves. After all, we are a team of chemists first. So, it is with those bona fides that we set out to develop the next generation of instrument that we would want to buy.
We hold these truths to be self-evident
When we are discussing photochemistry, these things we hold to be true:
Temperature matters. It continues to surprise us that much of the literature overlooks the effect of temperature on a photochemical reaction. If you submit a Pd cross coupling that didn’t proceed at room temperature, and that gave 95% yields “with added heat” without reporting the temperature you would likely not get that result published (or at least it shouldn’t). And yet this parameter is often completely ignored when discussing photochemistry.
Light intensity matters: If you double the amount of a catalyst, or base or substrate that you add to a reaction would expect both the yield and potentially the selectivity of a reaction to be greatly affected. And yet, the literature forces everyone to compare results from different light sources and intensity without discussion of the differences. We can’t recommend this piece enough (Ref 3). Photons are a reagent. This also isn’t a claim that more light is always better. But the right amount of light is an important parameter to scale up a photochemical reaction (Ref 4). For some (or even most) reactions the amount of light may not make much of a difference.Actually, it will likely always make a difference, we hold this truth less strongly.Be prepared for those that are.
Wavelength matters: There was a time when the majority of the literature reported reactions that were performed with either “blue” or “white” light. Fortunately, that has for the most part changed. But reaction schemes or figures that only list “blue” still cause us to die a little inside. Also, not all 450 nm LED spectra are equal. If you normally run your reaction at 450 nm, is the conversion better at 425 nm, 405 nm, 365 nm? If you aren’t at least curious about this …?
Photochemical Reactions scale up: The old myth that photochemistry doesn’t scale is simply a myth in not understanding what was happening in your reaction in the first place. Your reaction might be best in flow at scale. Or flow could be a flop and batch is best. Be prepared to do both.
One device should be able to do it all:
Often the correct experiment or parameter screen is not performed because it is prohibitively difficult to do the study. Do you want to survey catalysts, wavelengths, temperature and light intensity? It can end up being a pretty big experiment. Often, we are limited to picking only a few of those parameters to study to limit the work. For example, the selectivity displayed in this paper (open access) fascinates us (Ref 5). Is it temperature, wavelength, light intensity or all of the above? Innovation will be found in these subtle differences like these for the next generation of photochemical studies. The proper instrument to do this set of experiments doesn’t exist. Or at least it didn’t. We built it. One device to run a screen, scale up a reaction in batch and then try it in flow. This is the device that we want to use ourselves.
If you build it, they will come (Introducing the Lucent360)
We wanted a device suitable for both reproducible and repetition of a standard setup from day to day and flexible enough to quickly study wavelength, light and temperature from screen to scale. In both batch and flow. Why did we name our new reactor the Lucent360™ when the names PhotoRedox Box 2, PhotoRedox Box 1000 or Photoredox Box Plus names were still available? The simple answer is this. Lucent, means “glowing with light”. The reactor is a circle. Circles have 360 degrees. Did I mention that naming things is difficult?
The Lucent360’s flexible design gives you the best options to learn everything you need to know to take your photochemical reactions from screen to scale (batch and flow). Screen at a scale that best suits your needs, whether you are developing new methodology and need to test a large number of reagents and catalysts. Or maybe you want to run an optimized reaction in parallel on a large number of substrates. The Lucent360 affords you nearly unlimited flexibility in one device.
To start, choose a standard holder to screen up to 48 reactions (0.3 ml vials), 24 reactions (2 ml vials), 24 reactions (4 ml vials), 24 reactions (8 ml vials) or 12 reactions (20 ml vials) at once. The Lucent360 offers you your choice of wavelength, light intensity, (time) and temperature with control of the stirring. The Lucent360 contains 5 independently controlled and interchangeable high-powered LED modules available at 365 nm, 380 nm, 405 nm, 450 nm (425 nm, 530 nm, 650 nm, 740 nm to come?) offering you more light than any other commercial device (we have the actinometry data to prove it) in a safe enclosed environment. Accidently open the lid before your reactions are complete? The Lucent360 will safely shut off the lights before you are seeing stars.
Need more or less light? You have control over the light intensity to best control your reaction. (there’s so much power, 50% setting can be a good place to start) Connecting the Lucent360 to a standard heater/chiller unit enables accurate temperature-controlled reaction conditions (0° to 80° C) that are insulated from the heat of the light modules. Two temperature probes (included) can also connect to each of the Lucent360 vial holders, allowing for precise temperature measurements. The fill and flow rate of the reactor chamber is controlled by the manual valves accessed on the front panel. A supercharged built-in stir plate allows efficient stirring for each of your reactions. The reaction time can be controlled with an automatic shutdown of the lights and pumps. Or shut the lights off and let the pumps run to keep your samples at a constant temperature. So set it up and walk away. Need access to the chamber while your reactions are running? An easy access port allows the user to directly add reagents via syringe pump or add a gas line.
What really sets the Lucent360 apart from other photoreactors is the use of our multi-light screening vial holders. For example, with our 4 ml vial divided screening holder, in one experiment you can test four different wavelengths or light intensities simultaneously with 16 total reactions. Or in one experiment, have access to data for your favorite reaction with four catalyst concentrations at four wavelengths with temperature control), or one wavelength at four light intensities with four catalyst concentrations. How about a screen with four catalysts each at their optimal wavelength? Or mix and match. Go crazy, the possibilities are nearly endless. Find a configuration that you like best? Save the settings as 1 of the 5 available presets with a simple push of a button.
Need more material? Take your synthesis to a larger batch in the Lucent360 with the 700 ml reactor that fits directly into the instrument. The glass reactor is equipped with a Teflon seal, enables 100-700 ml reactions and includes a temperature probe to monitor the internal temperature, 3 ports for inlet/outlet of inert gas or addition/sampling of the reaction. The reactor includes two ports that enable the chiller to cool (or heat) the reaction to the appropriate temperature.
If you think your reaction is best suited for flow? Transition to the flow reactor that fits easily into the Lucent360. With 20 ml and 50 ml flow cell, turn your Lucent360 into a continuous production of your favorite material. An easy access port allows constant flow with external pumps into the temperature controlled, light controlled environment.
Still think the Lucent360 is more instrument than you need, then check out one of other photoredox boxes, here.
In the coming weeks, in addition to our regular irreverent look at the topics of interest in the photochemical field, we will be posting some the results that we have from the last few months with the Lucent360™. As more and more of the instruments ship out into the real world, we look forward to seeing what everyone out can dream up with the use of our instruments! We hope the Lucent360™ allows the study of photochemical reactions in ways that we haven’t even thought of and helps to extend the depths of understanding of the next wave of visible-light photochemistry.
References:
- Halperin, S. D.; Fan, H.; Chang, S.; Martin, R. E.; Britton, R. A Convenient Photocatalytic Fluorination of Unactivated C-H Bonds. Chemie – Int. Ed. 2014, 53 (18), 4690–4693. https://doi.org/10.1002/anie.201400420.
- DiRocco, D. A.; Dykstra, K.; Krska, S.; Vachal, P.; Conway, D. V.; Tudge, M. Late-Stage Functionalization of Biologically Active Heterocycles Through Photoredox Catalysis. Chem. Int. Ed. Engl. 2014, 53 (19), 4802–4806. https://doi.org/10.1002/anie.201402023.
- Bonfield, H. E.; Knauber, T.; Lévesque, F.; Moschetta, E. G.; Susanne, F.; Edwards, L. J. Photons as a 21st Century Reagent. Commun. 2020, 11: 804. https://doi.org/10.1038/s41467-019-13988-4.
- Harper, K. C.; Moschetta, E. G.; Bordawekar, S. V.; Wittenberger, S. J. A Laser Driven Flow Chemistry Platform for Scaling Photochemical Reactions with Visible Light. ACS Cent. Sci. 2019, 5 (1), 109–115. https://doi.org/10.1021/acscentsci.8b00728.
- John, A.; Paul, A.; Leitch, J. A.; Yamazaki, K.; Christensen, K. E.; Cassar, D. J.; Hamlin, T. A.; Dixon, D. J. Switchable, Reagent-Controlled Diastereodivergent Photocatalytic Carbocyclisation of Imine-Derived α-Amino Radicals. Chem. Int. Ed. 2021. ASAP https://doi.org/10.1002/anie.202107253.