
The Lucent360™ is the most complete photochemical reactor option available for controlling and screening reaction parameters like wavelength, light intensity, reaction time and temperature at both small and large scale. And while the Lucent360™ can do just about anything and everything photochemical in your lab, it’s also important to see how this this reactor can fit into the rest of your photochemical research and development program. A recent paper from GSK demonstrates how the Lucent360™ can help solve a key step during development in medicinal chemistry.
In their work entitled “Expedient Discovery of a Metallaphotoredox Cyanomethylation for Synthesizing α-Aryl Nitriles” lead author Gemma Cook and coworkers at GSK report how experiments with the Lucent360™ helped remove a highly toxic cyanide reagent from their synthesis while discovering a new reaction. Several marketed drugs include α-aryl nitriles, a valuable functional group for modulating interactions with targets of interest through bonding interactions while maintaining a small size. However, synthesis of these compounds often requires highly toxic cyanide reagents that can’t be used on industrial scale. Removing this toxic reagent would facilitate library screening of α-aryl nitriles compounds on a larger scale while also affording this functional group as synthetic handle for incorporating many other functional groups.
While attempting to alkylate something else photochemically with a standard Ir/Ni condition, the authors observed an unexpected cyanomethylation product from trapping of acetonitrile. This jump started development of a brand-new method for generating α-aryl nitriles. We won’t attempt to describe the full extent of the work at GSK in full (for that check out the link above) but this led to an extensive program using an HTE photoreactor and robotics to identify the optimal photocatalyst, nickel, ligand and base sources as well as the ideal stoichiometries lowering the loading of iridium and nickel without loss of yield.
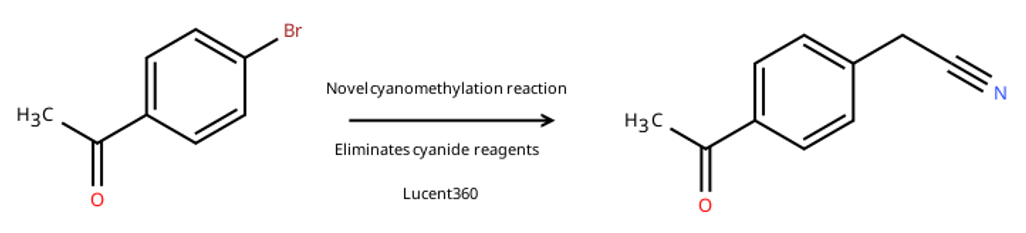
The optimal reaction parameters were used the Lucent360™ for a substrate scope with a wide selection of medicinally relevant functional groups. The authors used the Lucent360™ in the screening mode, using a vial holder containing 24x4ml vials with the samples irradiated by 5 independent light modules at 450 nm. The temperature of the reactions was held stable at 40 °C using the Lucent360 thermostatic bath and reactions were run for 22-88 hours. The authors demonstrated that the reaction was tolerate of a wide range of functional groups including amides, alcohols, phenols, sulfonamides and ethers wide modest but synthetically useful yields.

Finally, the authors applied their new method to the synthesis of a Senexin-derivative achieving a comparable yield for the cyanomethyl derivative in 1 step photochemically compared to 3 originally while also eliminating the use of a toxic cyanide reagent. The authors use the word “expedient” in the title of their paper to describe their work developing this new reaction which is quite apt as they have shown quickly you can turn a serendipitous observation into a tangible achievement if you have the right tools.
For more examples of the Lucent360:
Illuminating the Role of Alcohol Substrate in Nickel Photoredox Catalysis via Ground State and Transient Absorption Spectroscopy, Annika R. Holm, Rachel F. Wallick, Josh Vura-Weis, and Liviu M. Mirica, ACS Catalysis 2026 16 (2), 1522-1532
A unified approach to meta-selective methylation, mono-, di- and trifluoromethylation of arenes, Elisa Y. Lai, Lutz Ackermann and Magnus J. Johansson, Chemical Science, 2025, 16, 8478-8486

