We are huge fans of what could be classified as “creative photocatalysts”. One of our favorite papers over the past few years was this work that we wrote about here using Hypericum flowers as an organic dye for C-C bond formation. A few dried flower petals, a base and an LED and you have a new photochemical reaction. So, if your paper can be described as using a “Fenton Boat photocatalyst”, well you have our attention: These authors made a sustainable, useful photocatalyst out of three of the cheapest things available–wood, iron and sunlight.
If you Google “Fenton Boats”, you get links to a boat shop in Fenton, Michigan, but we’ll argue that soon you will get this recent paper in Angewandte from Zhijun Chen and coworkers entitled, “A Sustainable Wood-Based Iron Photocatalyst for Multiple Uses with Sunlight: Water Treatment and Radical Photopolymerization“
What’s a Fenton Boat? Stick around and we’ll explain. And show you a video of a photocatalyst boat.
While we had a little fun with that lead up that may make it seem that we are making light of this work, we’re not. This is truly impressive. The authors made a sustainable, useful photocatalyst out of three of the cheapest things available, wood, iron and sunlight. There are many big picture problems that can be aided with photocatalysis. But there are certain problems where the task is so vast like water treatment that the solution needs to be less than cheap to have any chance of making a realistic improvement. Few things are cheaper than wood, iron and sunlight.
A sustainable catalyst should be recyclable and derived from sustainable materials. Iron oxides are well known materials for heterogenous photocatalysis, although to be successful often need to be modified with photosensitizers or frameworks derived from fossil fuels. Wood is a natural replacement. Natural wood is fluorescent and phosphorescent due to lignin, cellulose and hemicellulose. For this work, modified wood doped with iron oxide acts as a photosensitizer for photocatalytic cycles, mainly photo-Fenton reactions and photopolymerizations.
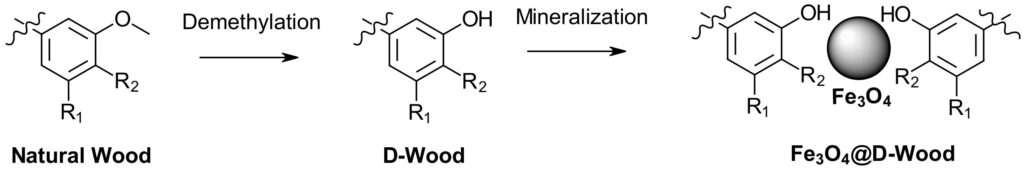
The catalyst is prepared by treating wood with BBr3 to remove the methyl groups from the lignin and creating free phenols. The D-Wood was then treated with FeCl3 followed by NaOH. This process was repeated 5 times, to give Fe3O4-D-Wood containing 19% weight percent Fe3O4 (Figure 1). This material was then extensively characterized, to an extent that we can’t do justice in this short piece. An alphabet soup of techniques characterized the structure including computer tomography (CT), X-ray diffraction (XRD), FT-IR, XPS, Electrochemical impedance spectroscopy (EIS), Cyclic voltammetry (CV) and visible absorption spectroscopy. All the data support a new material capable of proton electron transfer (PET) reactions consisting of a wood chamber filled with Fe3O4 particles coordinated by free phenols. The material has red-shifted absorbance observed after treating the D-Wood with iron confirming ligand to metal interactions. Fluorescence (1.64 ns) and Phosphorescence decay (0.38 ms) of D-Wood were confirmed demonstrating singlet and triplet states, while both were both suppressed upon addition iron, suggesting the new catalyst has non-emissive excited states. Similar characterization of Fe3O4-D-Wood (no demethylation) demonstrates the importance of the free phenols to catalytic activity.
Figure 1: Fe3O4-D-Wood photocatalyst
The authors next set up an experiment to test their new catalyst for the Photo-Fenton degradation of rhodamine B (RhB) with H2O2. In the dark, Fe3O4, Fe3O4-D-Wood, a mixture of separate iron and D-wood powders as a control and Fe3O4-D-Wood powder showed slow degradation (0.002-0.007 min-1). Turn on the artificial sunlight source and Fe3O4-D-Wood increases to 0.13 min-1 with 99% degradation of rhodamine by UV/Vis spectra while no increased rate was observed for the other species. Controls confirm that reaction is proceeding via the wood catalyst and not iron leaching into solution. Further experiments varying pH, testing stability of the catalyst and recycling the catalyst were all performed with a simulated pollution sample for the degradation of 8 common pollutants. After 40 minutes of irradiation, 51% of organic contaminants were removed.
As one does when making photocatalysts, the authors also tested materials made of different sources of natural wood including Beech wood, Pine wood, and Red Walnut with small variations in efficacy. Why did we feel the need to add this detail? Because for some reason the idea that different species of trees have different photochemical properties based on the structure of their lignin is fascinating to us simple chemists.
So, we promised you a Fenton Boat? Why a boat? Because small powder chunks of the wood catalyst sink to the bottom of a polluted body of water, limiting interaction with sunlight while a boat floats. The wooden boat was treated with BBr3/Fe conditions to generate a Fenton Boat that floats. The Fenton Boat absorbs polluted water via capillary action into the wood and concentrates in the pores and degrades via the Fenton reaction initiated by the photocatalyst. In a test experiment using artificial sunlight nearly 100% of RhB degradation was observed and in polluted water organic contaminants decreased by ~66%. The boat can be reused and more importantly recovered and moved by magnets. Just an incredible idea. There is far more work left to discuss including their experiments looking at radical traps, the kinetics, and discussion of mechanism, but we know everyone just wants to see the boat. For the purposes of scale, we’ll note that the boat fits inside a 1 L beaker. To clean up a larger body of water, we’re going to need a bigger boat.
Here is a video of the boat in action: